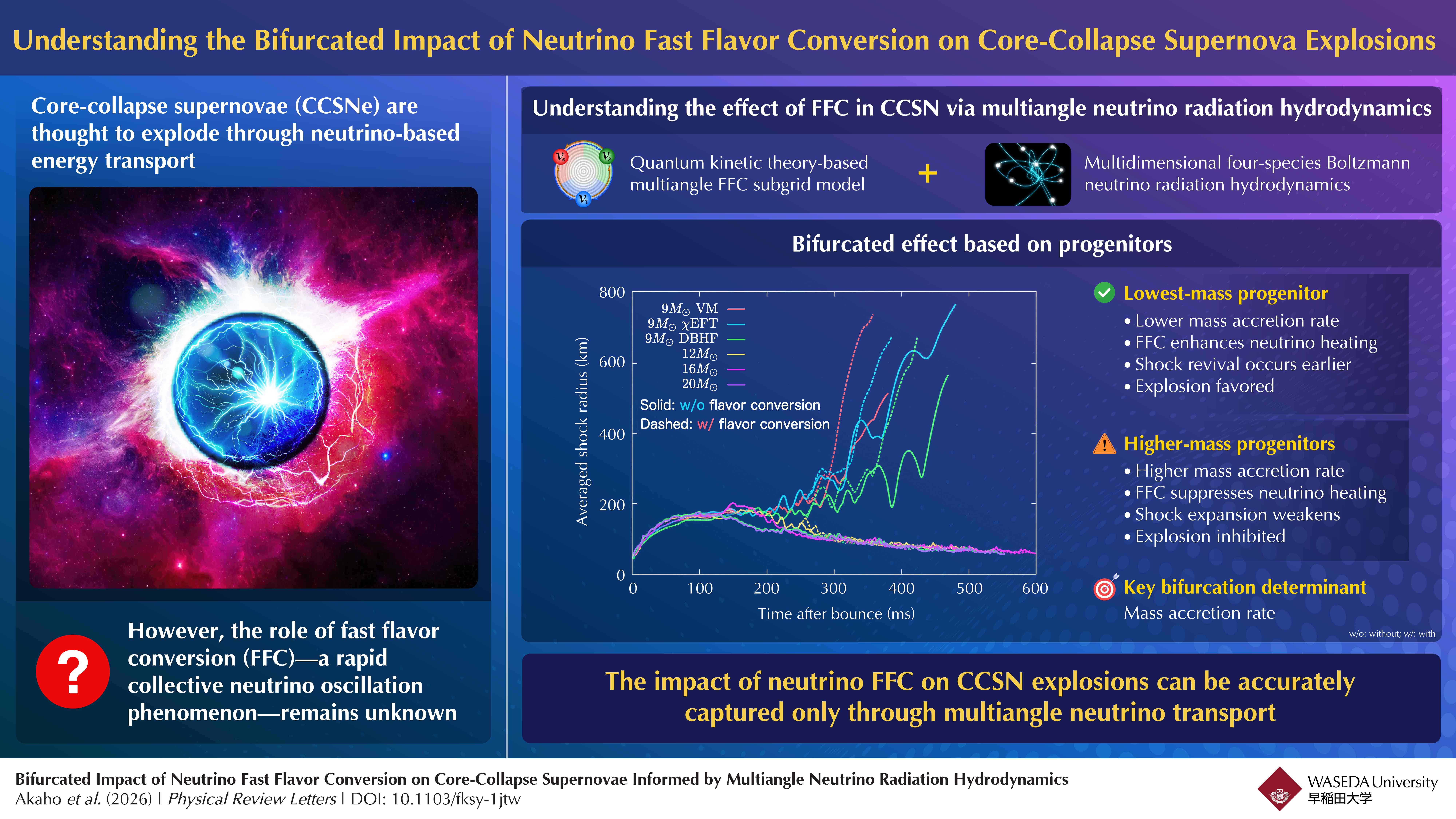
Core-collapse supernovae are thought to be powered by neutrino-driven energy transport. Now, researchers in Japan have shown that neutrino fast flavor conversion can either enhance or suppress supernova explosions depending on the progenitor star. Using advanced multiangle neutrino radiation hydrodynamics simulations, the team demonstrated that the mass accretion rate is the key factor governing this bifurcated behavior, offering new insight into the evolution and death of massive stars.
Our universe, filled with galaxies and stars, is full of mysteries. Over the centuries, astronomers have observed and documented supernova—the catastrophic explosion of stars—as some of the brightest and most energetic events in the universe. In particular, at the end of their lives, massive stars explode into core-collapse supernovae (CCSNe). Scientists believe these explosions to be mainly facilitated by neutrino-mediated energy transport. However, the effects of collective neutrino oscillations known as fast flavor conversion (FFC) on the CCSN explosion mechanism remain largely unclear.
Previous studies attempted to investigate the role of FFC in CCSNe using approximate “truncated moment” methods. However, these approaches cannot reliably capture the angular neutrino distributions needed to determine where FFC occurs.
A new study instead employs a multiangle treatment, allowing the researchers to directly model the angular behavior of neutrinos in momentum space.
The team of researchers, led by Assistant Professor/Junior Researcher Ryuichiro Akaho from the Faculty of Science and Engineering at Waseda University, Japan, along with co-authors Dr. Hiroki Nagakura from the National Astronomical Observatory of Japan and Professor Shoichi Yamada from Waseda University, has carried out CCSN simulations with multiangle neutrino transport to elucidate the impact of neutrino FFC on CCSNe.Their insightful findings were made available onlineon May 11, 2026, and have been published inVolume 136, Issue 19 ofthe journal Physical Review Letters on May 15, 2026. The paper was also selected as a “Featured in Physics” article by the journal editors, recognizing its significance and broad interest to the physics community.
In this study, the team combined a quantum kinetic theory-based FFC model with multidimensional Boltzmann neutrino radiation hydrodynamics simulations. Their framework directly identifies where FFC occurs using neutrino angular distributions calculated during the simulation itself. Akaho remarks: “We deploy our first-ever Boltzmann radiation hydrodynamics code that implements an FFC subgrid model, judge the occurrence of FFC directly from angular distributions obtained in simulations, and ascertain neutrino flavor states via physics-based quantum kinetic methodsimplemented through the Bhatnagar-Gross-Krook relaxation scheme. Crucially, we have already demonstrated this extended framework of neutrino transport in our previous work.”
The CCSN simulations presented in this study encompass successful as well as failed explosions, various progenitor models with zero-age main sequence masses of 9, 12, 16, and 20M⊙, and three different nuclear equations of state (EOSs), namely, variational method-based Furusawa-Togashi EOS, Dirac-Brückner-Hartree-Fock technique, and chiral effective field theory.
The researchers remarkably found that the impact of FFC on CCSN explosion is bifurcated depending on the progenitors. While FFC promotes shock revival and boosts the explosion energy for the lowest-massprogenitor, it has an inhibitory impact for higher-mass progenitors. The mass accretion rate is the main determinant governing this bifurcated effect. For a high value of mass accretion rate, the contribution of FFC to neutrino heating turns out to be negative,since the concurrent reduction in neutrino luminosity dominates over the enhancement of heating efficiency through FFC-driven spectral hardening of electron-type neutrinos. In contrast, FFC contribution to neutrino heating becomes positive foralow mass accretion rate.
“Our present results highlight the limitations of approximate neutrino transport and show that a multiangle treatment is essential for accurately capturing FFC effects. Otherwise, important FFC signals may be overlooked or even falsely identified,” highlights Akaho.
Overall, this work provides a robust argument for the involvement of neutrino FFC in the explosion mechanism of CCSNe, improving our understanding of the lifecycle of massive stars and potentially serving as a theoretical guide for future CCSN observations.